Crude oil pollution endangers humans, and the remediation of contaminated environments is an urgent undertaking[1].Crude oil can be classified into four fractions, namely, saturates, aromatics, resins, and asphaltenes(SARA)[2].Heavy oil contains a high percentage of resins and asphaltenes and generally shows low bioavailability on account of its low fluidity and high viscosity[3].Unfortunately, knowledge of the most effective functional microbes with stable degradation performance and the underlying mechanisms is lacking.
Bioremediation approaches are generally preferred over other types of pollution-management approaches, and over 200 types of bacteria are known to degrade crude oil components[4].The recent development and application of bacterial consortia to improve the efficiency of crude oil biodegradation have been reported[5-6].However, studies on the application of mixed microbial consortia to degrade crude oil containing heavy components are scarce.
The efficiency of crude oil degradation is strongly affected by external environmental parameters[7], including temperature, pH, and inorganic salt concentration.However, these parameters are often individualized for different bacteria.Thus, parameter optimization is crucial to enhance the synergistic degradation efficiency of microbial consortia.
In the present study, three microbial consortia with high oil-degradation efficiencies were isolated and enriched directly from heavy crude oil-contaminated soil.The effects of various environmental factors on the performance of the obtained crude oil-degrading microbial consortia were then determined.Finally, the structure of the enriched crude oil-degrading mixed microbial community was analyzed, the dominant functional species were investigated, and the relevant degradation mechanisms were explored.
1 Materials and Methods
1.1 Samples and media preparation
The crude oil used in this study was procured from Sinopec Group, Shandong, China.Three types of crude oil-contaminated soils were collected from the provinces of Shandong, Jiangsu, and Gansu, China.
The mineral salt medium(MSM), which contained 1.0 g/L K2HPO4, 1.0 g/L KH2PO4, 1.0 g/L NH4Cl, 0.5 g/L NaCl, 0.5 g/L MgSO4, 0.1 mg/L CaCl2, 0.1 mg/L FeSO4, 0.02 mg/L CuSO4, 0.02 mg/L ZnSO4, and 0.02 mg/L MnSO4, was adjusted to neutral pH.The nutrient broth medium contained 10.0 g/L peptone, 3.0 g/L beef extract, and 5.0 g/L sodium chloride(pH 7.2±0.2).The enriched medium was composed of a mixture of MSM and crude oil at a ratio of 200:1.The selective medium was composed of enriched medium amended with 15 g/L agar.
1.2 Crude oil-degrading consortium enrichment
Ten grams of each soil sample was transferred to a 250 mL conical flask and then added with 100 mL of enriched medium at 30 ℃ and 150 r/min.Each culture was supplemented with 0.5 g of crude oil every 10 d.After 50 d, the solution was centrifugated at 8 000 r/min for 5 min, and the cell pellet was collected.This pellet was resuspended.Then, 200 μL of the resuspended culture was spread onto the selective medium and cultured at 30 ℃ for 16 h.Microorganisms that grew on the selective medium were considered a member of the potential crude oil-degrading consortia.
1.3 Determination of biodegradation activity
Each microbial consortium was inoculated in sterilized nutrient broth medium amended with 0.5% crude oil for proliferation until the OD600(optical density for the suspension at 600 nm)of the solution reached 0.6.Next, 5 mL of the culture, 95 mL of MSM, and 0.5 g of crude oil were added to flasks and incubated at 150 r/min.Triplicate flasks were established and sampled every 3 d.The control with no consortium inoculation was used to quantify the abiotic oil loss.The residual crude oil was measured using the gravimetric method[8].The percentage of crude oil degraded was calculated as follows:
(1)
where m0 is the mass of the original crude oil and m1 is the mass of the remaining oil after incubation.
The crude oil was separated, and changes in its SARA fraction were determined by column chromatography according to the Chinese industrial standard SY/T 5119—2016[9].
1.4 Determination of optimal environmental conditions for biodegradation
The pH of the reaction media was adjusted to 6.0 to 8.0, and incubation temperatures of 20 to 40 ℃ were tested to assess their effects on crude oil biodegradation at the optimal pH.The inorganic salt concentration was adjusted to 0.8% to 12% while maintaining the same proportion of components described in MSM.For each reaction, the remaining oil in the flask was measured by the gravimetric method.
1.5 Analysis of the microbial consortium structure
The incubation solution was collected and passed through 0.22 μm filters(Millipore, USA).The filters were then subjected to DNA extraction by using a Qiagen DNA Kit.Polymerase chain reaction was performed to amplify the 16S rRNA gene using the bacterial primers 338F(5’-ACTCCTACGGGAGGCAGCAG-3’)and 806R(5’-GGACTACHVGGGTWTCTAAT-3’).A DNA library was constructed and loaded onto the Illumina Miseq PE300 platform at Majorbio Bio-pharm Technology Co., Ltd.(Shanghai, China).
2 Results and Discussion
2.1 Development of the crude oil-degrading consortia
The three microbial consortia were named GS, JS, and SD according to their retrieval sources.These consortia were further mixed at a biomass ratio of 1:1:1 to form a new functional microbial consortium.Each functional microbial consortium was inoculated, and the crude oil-biodegradation activity of each sample was determined.After incubation, oil droplets formed and dispersed in the liquid phase of the flask, and the oil sheen on the surface of the medium faded(see Fig.1).The appearance of dispersed oil droplets suggests the presence of biodegradation activity.Such phenomena were not observed in the control, which revealed a total oil loss rate of only approximately 6.1% after 30 d of incubation.The overall oil-degradation rates of the consortia GS, JS, and SD and their mixture after 30 d of incubation were(31.4±3.2)%,(26.4±2.8)%,(29.4±1.1)%, and(48.0±3.5)%, respectively(see Fig.2).Therefore, all retrieved consortia display oil-degradation potential, with their mixture possessing the highest oil-degradation ability.The optimal ability of the consortium mixture to degrade heavy crude oil efficiently may be attributed to microbial cooperation and synergy, which could increase extensive enzymatic capacities for the utilization of oil.

(a)

(b)
Fig.1 Crude oil-containing flasks for 30 d at 30 ℃ with the microbial consortia.(a)Before incubation;(b)After incubation
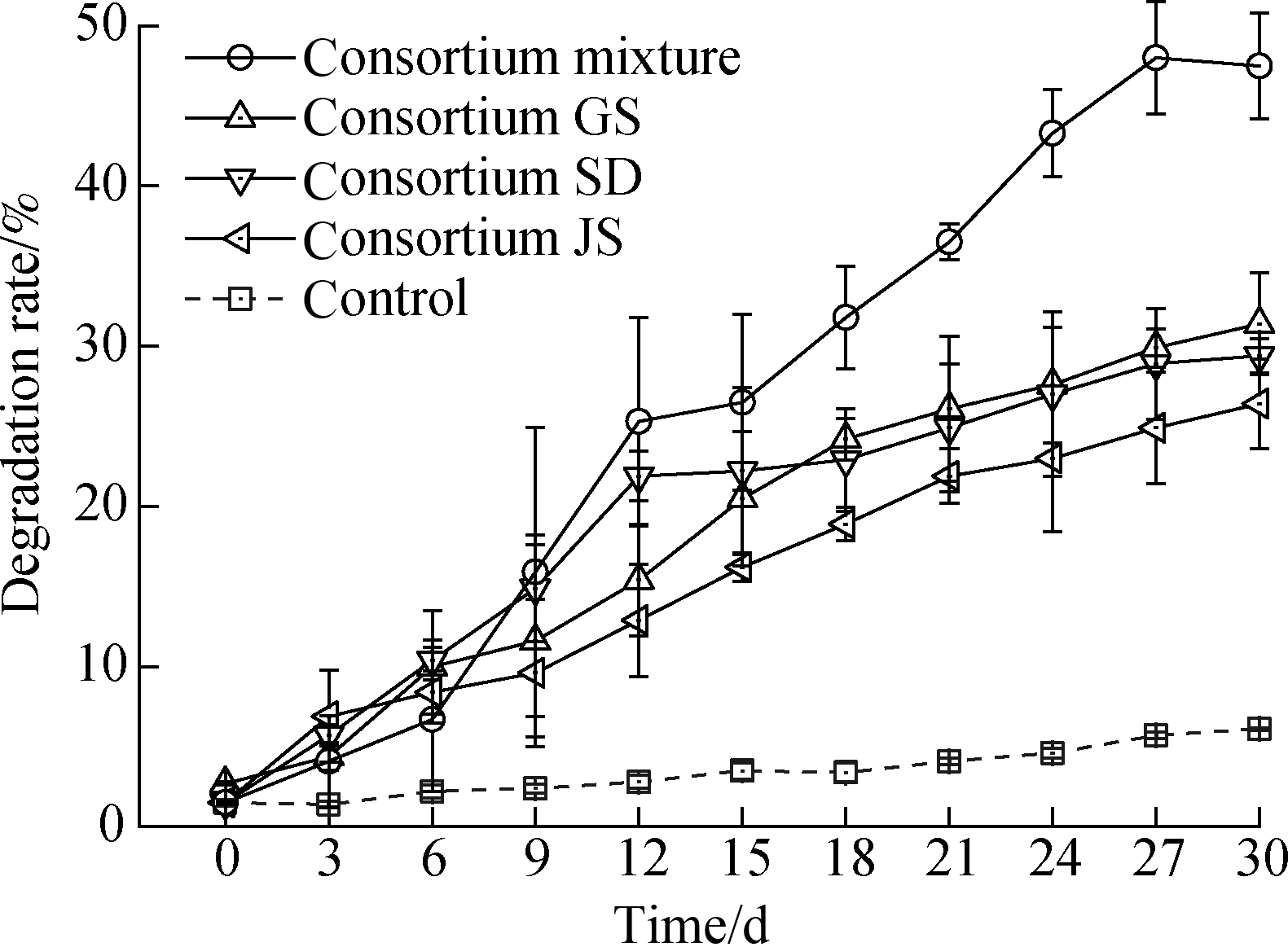
Fig.2 Crude oil-degradation rate curves of the microbial consortia GS, JS, and SD and their mixture
2.2 Degradation efficiencies for crude oil SARA fractions
Changes in the SARA fractions of the crude oil were monitored, and results indicated that saturates, aromatics, and resins, but notasphaltenes, were efficiently degraded by the consortium mixture(see Fig.3).The saturated hydrocarbon level in the flasks continuously decreased over the course of incubation by 79.7%.The concentration of aromatics declined by 35.9% within the first 15 d of incubation, but only another 9.7% reduction was achieved from day 15 to day 30.The resin content remained similar to the initial mass within the first 15 d of incubation and decreased by 11.1% from day 15 to day 30.
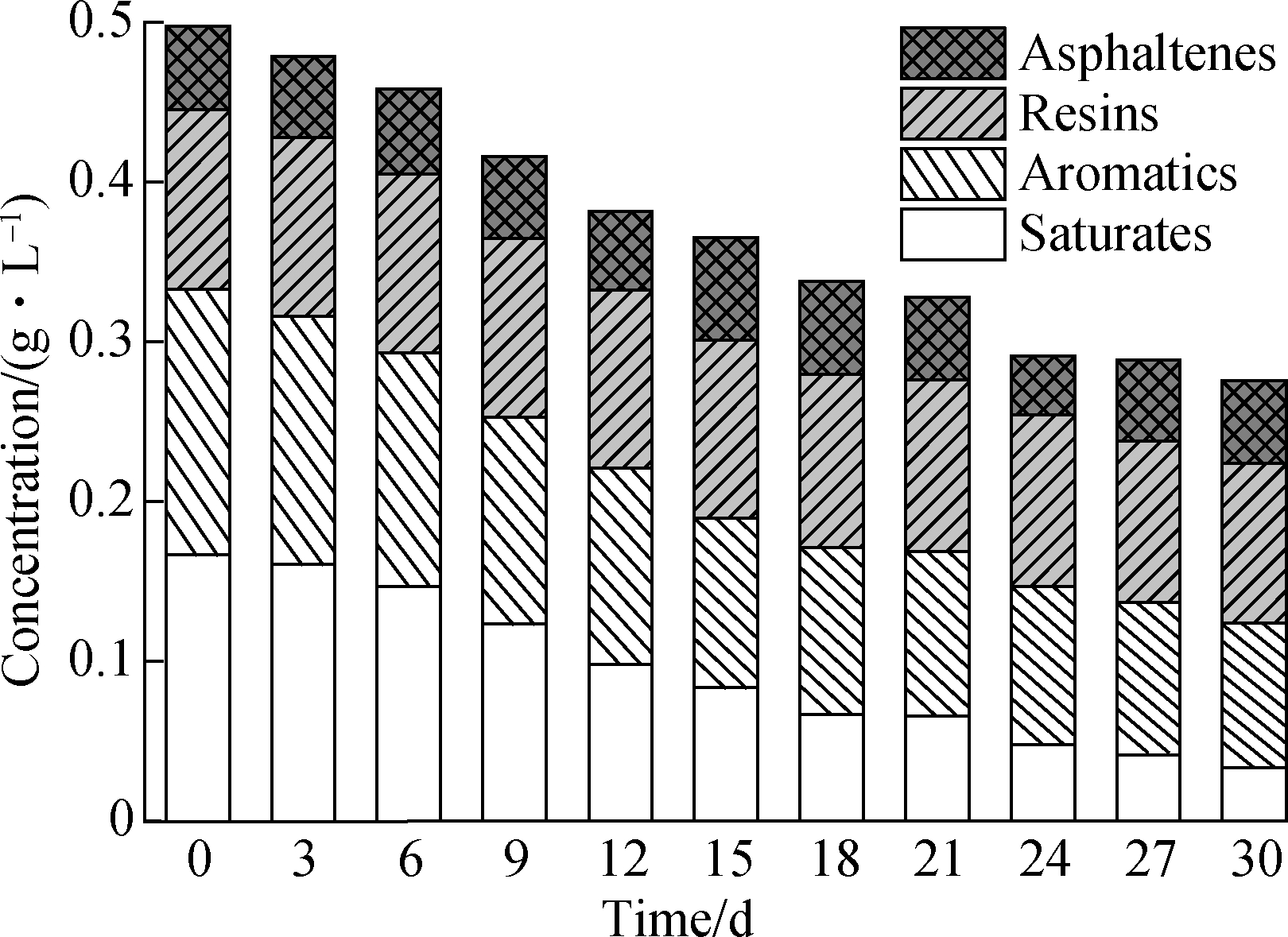
Fig.3 Changes in the crude oil SARA(saturates, aromatics, resins, and asphaltenes)fractions during biodegradation by the consortium mixture
The above results confirm that the consortium mixture possesses a promising ability to degrade heavy crude oil but not asphaltenes.Because of their simple structures and low hydrophobicity, saturated hydrocarbons in crude oil are preferentially degraded[10].When mononuclear aromatics and PAHs with low molecular weights are degraded, the residuals of the aromatics are generally thermodynamically stable and fairly refractory[11], leading to a low degradation rate from day 15 to day 30 of the incubation period.
The biodegradation of resins andasphaltenes in this work was very slow and time consuming, likely because of the high viscosity and density and poor fluidity of these substances[12].Some evidence suggests that the molecular structures of these molecules could change during incubation but without complete mineralization[13].The slight decrease in the mass of resins and the stability of asphaltenes indicate that these refractory components may eventually be biodegraded but require a longer incubation time.
2.3 Optimal environmental conditions for crude oil biodegradation
The consortium mixture was cultured under different reaction conditions for 10 d, and biodegradation efficiency tests indicated that high crude oil-biodegradation efficiency((10.4±2.8)%)could be obtained under neutral pH conditions(see Fig.4(a)).An incubation temperature of 30 ℃ was verified to be optimal for crude oil degradation, and the highest degradation rate((19.6±3.3)%)was obtained under this temperature(see Fig.4(b)).A low range of mineral salt concentrations(0.8% to 4.0%)exerted negligible effects on the crude oil-degradation efficiency of the consortium mixture(see Fig.4(c)).However, a lower crude oil-biodegradation rate of(2.3±1.0)% was obtained when the salt concentration reached twice the original value.

(a)

(b)

(c)
Fig.4 Crude oil-degradation rates of the consortium mixture after 10 d of incubation.(a)Different pH values;(b)Different temperatures;(c)Different mineral salt concentrations
Neutral conditions have been verified to benefit the biodegradation process.The consortium in this work was sensitive to pH changes, likely because biological processes, such as enzymatic reactions and the utilization of electron acceptors and nutrients, are affected by pH[14].Changes in temperature could change the solubility, viscosity, diffusion, and transformation of hydrocarbons, thereby affecting the bioavailability of oil[15].High salinity inhibited the consortium’s biodegradation efficiency, likely because this condition leads to unfavorably high osmotic pressures and altered sorption of toxic or essential ions[16].
2.4 Variations in microbial community diversity during incubation
The richness and diversity indices of the consortium mixture communities before and after incubation were analyzed.The observed OTU number of the day-30 sample was close to that of the day-0 sample.No significant difference between the ACE indices of these samples was noted(P=0.87).The Shannon and Simpson indices of the day-30 sample were not significantly different from those of the day-0 sample(P>0.05; see Tab.1).
Tab.1 Species richness and diversity indices of different samples at the OTU level
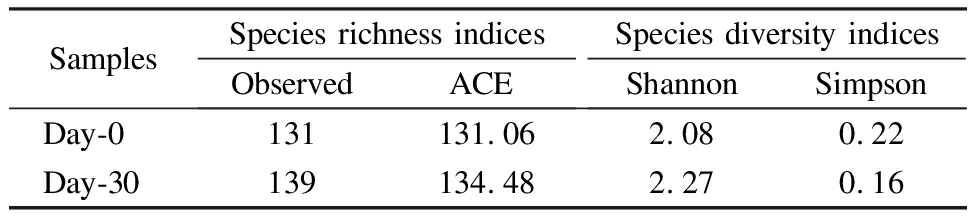
SamplesSpecies richness indicesObservedACESpecies diversity indicesShannonSimpsonDay-0131131.062.080.22Day-30139134.482.270.16
The genera Pseudomonas, Stenotrophomonas, Brucella, Serratia, Brevundimonas, Achromobacter, Macellibacteroides, and Citrobacter were dominant(see Fig.5).Whereas the abundances of Pseudomonas, Stenotrophomonas, and Brevundimonas decreased by 24.7%, 11.2%, and 2.4%, respectively, after 30 d of culture, the proportions of Brucella, Serratia, Achromobacter, and Macellibacteroides increased from the day-0 sample to the day-30 sample.
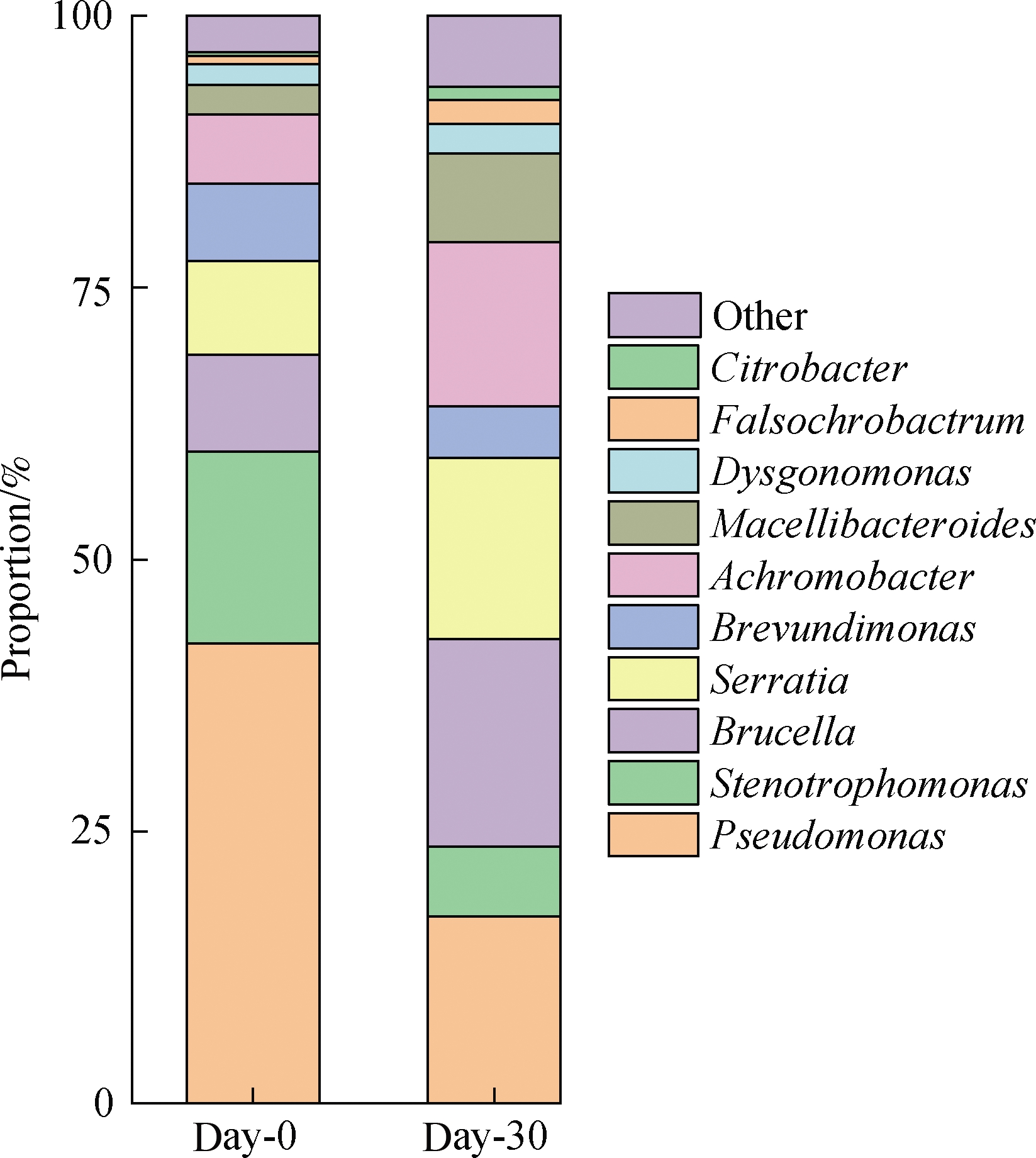
Fig.5 Dominant genera in the consortium mixture
The relatively stable microbial composition observed in the mixed consortium in this study suggests that the dominant species in the enriched consortium play important roles in crude oil degradation.Pseudomonas is widely reported to biodegrade saturates efficiently, and some members of this genus could produce biosurfactants to increase the bioavailability of oil components[17].Stenotrophomonas, Citrobacter and Brevundimonas could degrade some aromatics[18-19].Increases in the proportions of Brucella, Serratia, and Achromobacter could be related to the cleavage and modification processes of PAHs and aromatic cores in resins[20-21].Although Macellibacteroides is rarely mentioned in the biodegradation of crude oil, it has a close phylogenetic relationship with Dysgonomonas(see Fig.6), which could degrade phenol and dimethylphenol[22].The phylogenetic tree also showed that some dominant genera, such as Pseudomonas, Brucella, and Serratia, are not close to each other in terms of evolutionary relationships.This finding suggests the importance of the cooperation of multiple functional microbes for crude oil degradation.Therefore, enhancements in the crude oil-degradation activities of the mixed consortium may probably be attributed to the increase in oil bioavailability and cooperation of diverse microbial species in multiple biological metabolic pathways.
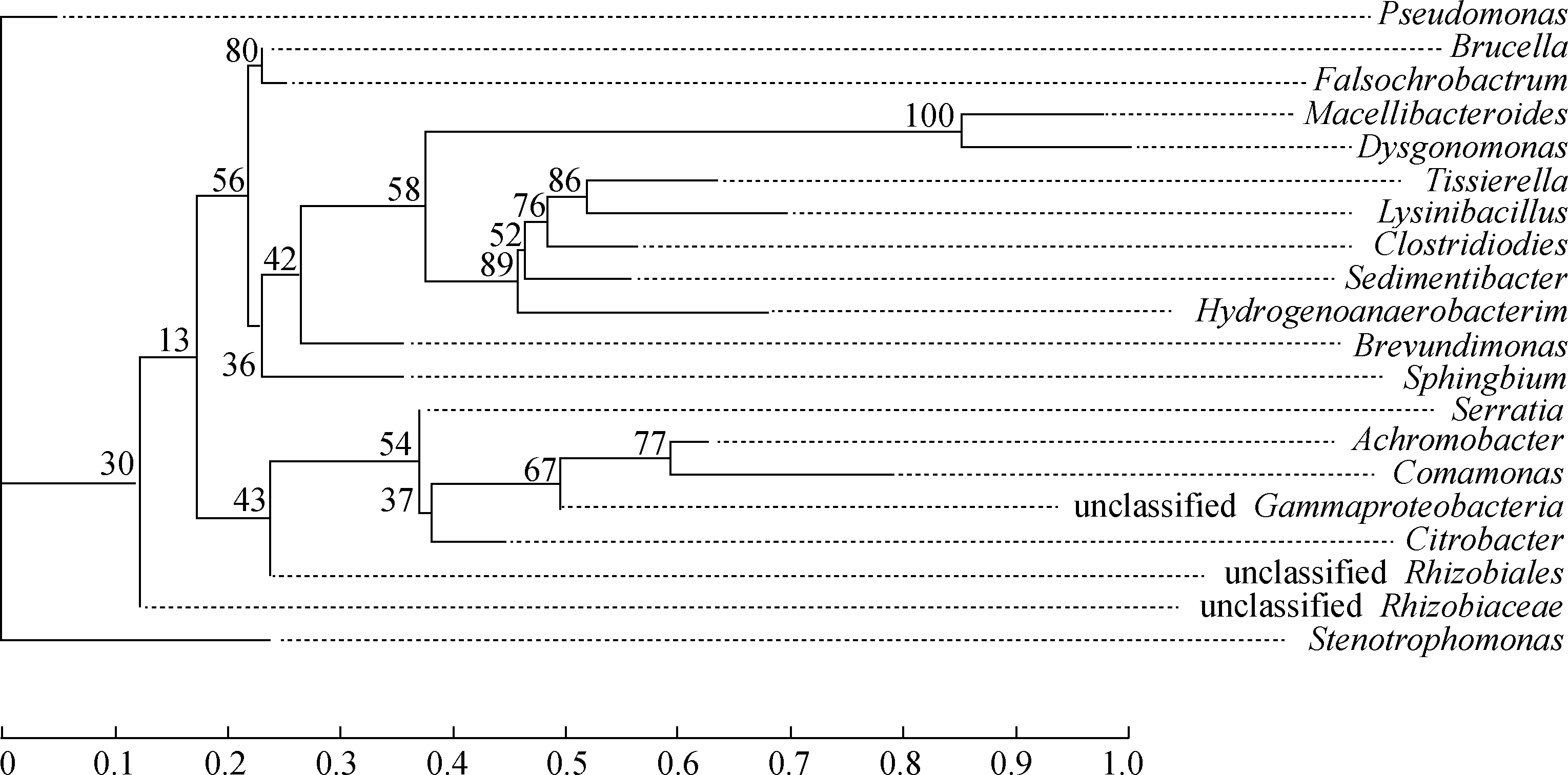
Fig.6 Phylogenetic tree of the dominant species of the consortium mixture(including the top 20 species in terms of total abundance at the genus level)
3 Conclusions
1)All of the enriched and isolated consortia could biodegrade crude oil.Among the samples tested, the consortium mixture showed the highest degradation rate((48.0±3.5)%)after 30 d of incubation.The consortium mixture could degrade 79.7% of the saturated hydrocarbons, 45.7% of the aromatics, and 11.1% of the resins in crude oil; asphaltenes were not observably degraded by this sample.
2)A neutral pH, culture temperature of 30 ℃, and salinity of no more than double the original MSM concentration could promote crude oil biodegradation.
3)The dominant genera of the consortium mixture were Pseudomonas, Stenotrophomonas, Brucella, Serratia, Brevundimonas, and Achromobacter.The proportions of these bacteria showed some variations over the course of incubation, but the richness and diversity of the microbial communities were stable.
[1] Singh H,Bhardwaj N, Arya S K, et al.Environmental impacts of oil spills and their remediation by magnetic nanomaterials[J].Environmental Nanotechnology, Monitoring and Management, 2020, 14: 100305.DOI: 10.1016/j.enmm.2020.100305.
[2] Obafemi Y D, Taiwo O S, Omodara O J, et al.Biodegradation of crude petroleum by bacterial consortia from oil-contaminated soils in Ota, Ogun State, South-Western, Nigeria[J].Environmental Technology and Innovation 2018, 12: 230-242.DOI: 10.1016/j.eti.2018.09.006.
[3] Xue J, Yu Y, Bai Y, et al.Marine oil-degrading microorganisms and biodegradation process of petroleum hydrocarbon in marine environments: A review[J].Current Microbiology, 2015, 71(2): 220-228.DOI: 10.1007/s00284-015-0825-7.
[4] Parach A, Rezvani A, Assadi M M, et al.Biodegradation of heavy crude oil using Persian Gulf autochthonous bacterium[J].International Journal of Environmental Research, 2017, 11(5): 667-675.DOI: 10.1007/s41742-017-0059-6.
[5] Cui J, Huang L, Wang W, et al.Maximization of the petroleum biodegradation using a synthetic bacterial consortium based on minimal value algorithm[J].International Biodeterioration and Biodegradation, 2020, 150: 104964.DOI: 10.1016/j.ibiod.2020.104964.
[6] Poi G,Aburto-Medina A, Mok P C, et al.Large scale bioaugmentation of soil contaminated with petroleum hydrocarbons using a mixed microbial consortium[J].Ecological Engineering, 2017, 102: 64-71.DOI: 10.1016/j.ecoleng.2017.01.048.
[7] Liu Y, Li C, Huang L, et al.Combination of a crude oil-degrading bacterial consortium under the guidance of strain tolerance and a pilot-scale degradation test[J].Chinese Journal of Chemical Engineering, 2017, 25(12): 1838-1846.DOI: 10.1016/j.cjche.2017.02.001.
[8] Khanpour-Alikelayeh E, Partovinia A, Talebi A, et al.Investigation of Bacillus licheniformis in the biodegradation of Iranian heavy crude oil: A two-stage sequential approach containing factor-screening and optimization[J].Ecotoxicology and Environmental Safety, 2020, 205: 111103.DOI: 10.1016/j.ecoenv.2020.111103.
[9] National Energy Administration.Ananlysis method for family composition of rock extract and crude oil:SY/T 5119—2016[S].Beijing: Petroleum Industry Press, 2016.(in Chinese)
[10] Chen W, Kong Y, Li J, et al.Enhanced biodegradation of crude oil by constructed bacterial consortium comprising salt-tolerant petroleum degraders and biosurfactant producers[J].International Biodeterioration and Biodegradation, 2020, 154: 105047.DOI: 10.1016/j.ibiod.2020.105047.
[11] Wang X,Cai T, Wen W, et al.Surfactin for enhanced removal of aromatic hydrocarbons during biodegradation of crude oil[J].Fuel, 2020, 267: 117272.DOI: 10.1016/j.fuel.2020.117272.
[12] Minai-Tehrani D, Rohanifar P, Azami S.Assessment of bioremediation of aliphatic, aromatic, resin, and asphaltene fractions of oil-sludge-contaminated soil[J].International Journal of Environmental Science and Technology,2015, 12(4): 1253.DOI: 10.1007/s13762-014-0720-y.
[13] Kopytov M A, Filatov D A, Altunina L K.Biodegradation of high-molecular-mass heteroatomic components of heavy oil[J].Petroleum Chemistry, 2014, 54(1): 58-64.DOI: 10.1134/S0965544113040087.
[14] Liu B, Liu J,Ju M, et al.Purification and characterization of biosurfactant produced by Bacillus licheniformis Y-1 and its application in remediation of petroleum contaminated soil[J].Marine Pollution Bulletin, 2016, 107(1): 46-51.DOI: 10.1016/j.marpolbul.2016.04.025.
[15] Al-Hawash A B,Dragh M A, Li S, et al.Principles of microbial degradation of petroleum hydrocarbons in the environment[J].The Egyptian Journal of Aquatic Research, 2018, 44(2): 71-76.DOI: 10.1016/j.ejar.2018.06.001.
[16] Qin X, Tang J C, Li D S, et al.Effect of salinity on the bioremediation of petroleum hydrocarbons in a saline-alkaline soil[J].Letters in Applied Microbiology, 2012, 55(3): 210-217.DOI: 10.1111/j.1472-765X.2012.03280.x.
[17] Zhang X,Xu D, Zhu C, et al.Isolation and identification of biosurfactant producing and crude oil degrading Pseudomonas aeruginosa strains[J].Chemical Engineering Journal, 2012, 209: 138-146.DOI: 10.1016/j.cej.2012.07.110.
[18] Tiwari B, Manickam N, Kumari S, et al.Biodegradation and dissolution of polyaromatic hydrocarbons by Stenotrophomonas sp.[J].Bioresource Technology, 2016, 216: 1102-1105.DOI: 10.1016/j.biortech.2016.06.047.
[19] Witono B.Biodegradation of used synthetic lubricating oil by Brevundimonas diminuta AKL 1.6[J].Makara Journal of Science, 2017, 21(3): 136-142.DOI: 10.7454/mss.v21i3.7382.
[20] Muthukumar N, Mohanan S, Maruthamuthu S, et al.Role of Brucella sp.and Gallionella sp.in oil degradation and corrosion[J].Electrochemistry Communications, 2003, 5(5): 421-425.DOI: 10.1016/S1388-2481(03)00093-6.
[21] Huang Y, Zhou H,Zheng G, et al.Isolation and characterization of biosurfactant-producing Serratia marcescens ZCF25 from oil sludge and application to bioremediation[J].Environmental Science and Pollution Research, 2020, 27(22): 27762.DOI: 10.1007/s11356-020-09006-6.
[22] Li X, Zhao L, Adam M.Biodegradation of marine crude oil pollution using a salt-tolerant bacterial consortium isolated from Bohai Bay, China[J].Marine Pollution Bulletin.2016, 105(1): 43-50.DOI: 10.1016/j.marpolbul.2016.02.073.